 
The numbers alongside the traces refer to the nanomoles of NADH oxidized per minute per milligram of protein. In the case of trace c, MAL was added at the time indicated by the arrow. A, Mitochondria (0.05 mg of protein) were incubated in 2 mL of the standard medium containing 10 EU PH-MDH then, 4 μ m NADH was added, and the fluorescence (λ ex 340 nm λ em 456 nm) was continuously monitored (trace a). The numbers alongside the traces refer to the nanomoles of NADH oxidized per minute per milligram of protein.įluorimetric measurement of NADH oxidation rate at low NADH concentrations by the external dehydrogenase and by MAL/OAA shuttle in DWM. DHAP (5 μ m) was also added to test the revealing system. DWM and PCM were treated as in A and A', but the reaction was started by the additions of 0.5 EU α-glycerophosphate dehydrogenase (α-GPDH) and 10 m m α-GP. C and C', Reconstruction of the α-GP/DHAP shuttle. Then, 10 μ m Asp and 1 m m aminooxyacetate were added to test the revealing system and to inhibit the transaminase, respectively. 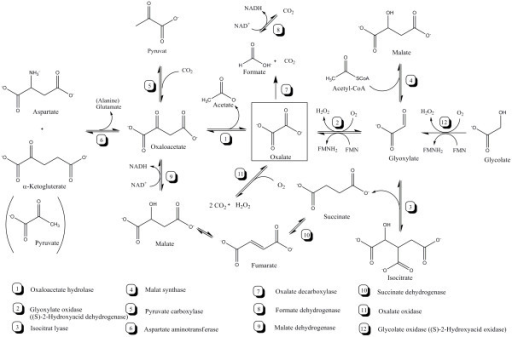
This was carried out as above, but 0.5 m m 2-oxoglutarate, 0.5 m m Glu, and 0.5 EU Asp aminotransferase (AAT) were also added to mitochondria. B and B', Reconstruction of the MAL/Asp shuttle. Where indicated, Phesucc, 10 m m in DWM and 2 m m in PCM, was also present in the medium. DWM and PCM (0.1 mg of protein) were incubated at 25☌ in 2 mL of the standard medium containing 0.3 m mannitol, 5 m m MgCl 2, 10 m m KCl, 1 m m ADP, 10 m m K-phosphate (pH 7.20), and 0.1% (w/v) defatted bovine serum albumin (BSA) addition was also made of 0.2 m m NADH and 10 m m EDTA plus 10 m m EGTA, which inhibit the NADH DH Ext the residual activity of NADH DH Ext was zeroed as reported in “Materials and Methods.” The reaction was started by adding 0.5 EU PH-MDH and 10 m m MAL, and the A 340 was continuously monitored. A and A', Reconstruction of the MAL/OAA shuttle. The existence of a MAL/OAA shuttle and the lack of both MAL/Asp and α-GP/DHAP shuttles in DWM (A-C) and PCM (A'-C'). The NADH DHExt contribution to NADH oxidation increased with increasing NADH concentration. We determined the kinetic parameters of the enzymes and the antiporter involved in NADH oxidation, and, on the basis of a kinetic analysis, we showed that, at low physiological NADH concentrations, oxidation via the MAL/OAA shuttle occurred with a higher efficiency than that due to the NADH DHExt (about 100- and 10-fold at 1 microm NADH in durum wheat mitochondria and in potato cell mitochondria, respectively). No NAD(P)H oxidation occurred arising from the MAL/Aspartate and the alpha-glycerophosphate/dihydroxyacetonphosphate shuttles. In durum wheat mitochondria and potato cell mitochondria, the rate of NADH oxidation was limited by the rate of a novel carrier, the MAL/OAA antiporter, which is different from other carriers thought to transport OAA across the mitochondrial membrane. 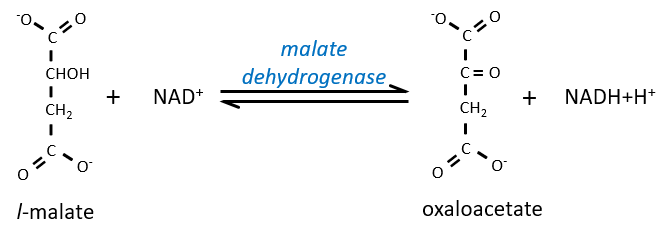
We found that external NADH and NADPH can be oxidized via two separate DHExt, whereas under conditions in which the activities of NAD(P)H DHExt are largely prevented, NADH (but not NADPH) is oxidized in the presence of external malate (MAL) and MAL dehydrogenase, in a manner sensitive to several non-penetrant compounds according to the occurrence of the MAL/oxaloacetate (OAA) shuttle. We investigated whether and how mitochondria from durum wheat (Triticum durum Desf.) and potato (Solanum tuberosum), isolated from etiolated shoots and a cell suspension culture, respectively, oxidize externally added NADH via the mitochondrial shuttles in particular, we compared the shuttles and the external NADH dehydrogenase (NADH DHExt) with respect to their capacity to oxidize external NADH. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
